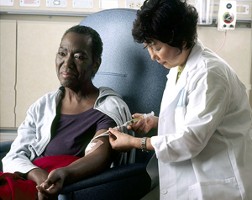
The U.S. Food and Drug Administration (FDA) has approved the first clinical trial in humans of brightly glowing nanoparticles to light up cancer cells to aid in diagnosing and treating cancer. The trial with five melanoma patients at Memorial Sloan-Kettering Cancer Center (MSKCC) in New York City will test if the technology is safe and effective in humans.
The nano particles, called Cornell Dots for the university that conducted the research, are silica spheres less than 8 nanometers in diameter that enclose several dye molecules. (A nanometer is one-billionth of a meter.) The silica shell, essentially glass, is chemically inert and small enough to pass through the body and out in the urine. For clinical applications, the dots are coated with polyethylene glycol so the body will not recognize them as foreign substances.
To make the dots stick to tumor cells, organic molecules that bind to tumor surfaces or specific locations within tumors can be attached to the shell. When exposed to near-infrared light, the dots glow much brighter than the unencapsulated dye, which serves as a beacon to identify the target cells.
For the human trials, the dots will be labeled with radioactive iodine, which makes them visible in PET scans to show how many dots are taken up by tumors and where else in the body they go and for how long.
The researchers say this technology can show the extent of a tumor’s blood vessels, cell death, treatment response, and invasive or metastatic spread to lymph nodes and distant organs. Studies on mice at MSKCC have already indicated that the technology is safe and can be cleared from the body by the kidneys.
Read more: Research Institutes Collaborate on Cancer Nanomedicine
* * *

 RSS - Posts
RSS - Posts
[…] This post was mentioned on Twitter by Alltop Science, Alan Kotok. Alan Kotok said: FDA Approves Trial of Nanotech Cancer Cell Markers | #Science #Business http://t.co/AMdXLuS #ScienceBusiness […]
I have been diagnozed with Testicular cancer. Is there any current medical trials for such patients with regard to nanomedicine?
Mr. du Preez:
ClinicalTrials.gov lists 88 trials currently dealing with testicular cancer that are recruiting for subjects …
http://www.clinicaltrials.gov/ct2/results?term=testicular&recr=Open
You and your doctor can review the list and select the ones that apply most directly to your case. Good luck.
– Alan
[…] http://sciencebusiness.technewslit.com/?p=2990 […]
I have been diagnosed with metastatic Merkel cell carcinoma and am extremely interested in nanotechnology treatment.
Thank you Mr. Beck. The principal investigator for this clinical trial is Dr. Michelle Bradbury at Memorial Sloan-Kettering Cancer Center in New York. You can reach Dr. Bradbury at 646-888-3373. Here’s her Web page: http://www.mskcc.org/cancer-care/doctor/michelle-bradbury . Good luck – AK