19 September 2014. The pharmaceutical company Boehringer Ingelheim is licensing from biopharmaceutical enterprise CureVac a vaccine based on RNA to generate an immune response for treating lung cancer. The deal has a total potential value to CureVac of €465 million ($US 597 million).
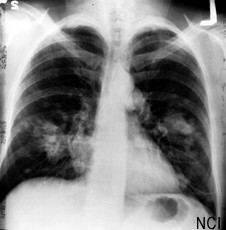
CureVac, in Tübingen, Germany, develops vaccines and treatments based on messenger RNA, or mRNA, that translates the genetic code in DNA to proteins expressed through an individual’s genes. The company’s technology harnesses mRNA to generate an immune response, either as a vaccine to prevent infectious diseases or, in the case of cancer, to attack cancer cells. CureVac has immunotherapies in early- and intermediate-stage clinical trials to treat prostate and non-small cell lung cancer, the most common form of lung cancer.
Boehringer Ingelheim, in Ingelheim, Germany, offers afatinib as a treatment for non-small cell lung cancer on the market in the U.S., Europe, and Japan marketed under the brand names Giotrif or Gilotrif. The company also has drug candidates to treat colorectal and non-small cell lung cancer, as well as acute myeloid leukemia, in late-stage clinical trials.
Under the deal, Boehringer Ingelheim gains exclusive worldwide rights to develop and commercialize CureVac’s CV9202 vaccine now in an early-stage clinical trial as a treatment for non-small cell lung cancer. The underlying technology to design CV9202, known as RNActive, remains part of CureVac’s intellectual property.
Boehringer Ingelheim plans to test CV9202 as a supplement to its afatinib drug for treating some types of advanced or metastatic non-small cell lung cancer, or as a supplement to chemo- and radiation therapy to treat inoperable forms of the disease. The agreement gives CureVac a one-time payment of €35 million ($45 million), and eligibility for up to €430 million ($552 million) in milestone payments and royalties on sales.
Read more:
- Early Clinical Trial to Test Leukemia Antibody Safety
- Immunotherapy Start-Up Adds $134M in Venture Funding
- Immunocore, Lilly Collaborate on T-Cell Cancer Therapies
- Personal Leukemia Immunotherapy Given Breakthrough Status
- Engineered Antibody Given FDA Breakthrough Tag for Myeloma
* * *

 RSS - Posts
RSS - Posts
You must be logged in to post a comment.