3 November 2015. A collaboration of engineering and medical researchers is developing an implantable artificial kidney to reduce the need for dialysis in people with kidney failure awaiting a transplant. The four-year project combining teams from University of California in San Francisco with Vanderbilt University in Nashville, is funded by a $6 million grant from National Institute of Biomedical Imaging and Bioengineering, part of National Institutes of Health.
The occurrence of kidney failure — also known as end-stage renal disease — is increasing in the U.S., as the population ages and becomes heavier, leading to high blood pressure and diabetes, the two leading causes of kidney disease. According to National Kidney Foundation, some 26 million people in the U.S. have kidney disease, leading to 47,000 deaths per year, making it the 9th leading cause of death. Men, African-Americans, and Hispanic-Americans are more likely to get kidney disease.
Once kidneys fail, dialysis or transplant is needed. National Kidney Foundation says about 450,000 people in the U.S. are on dialysis, requiring 3 visits a week for hours at a time. In addition, some 185,000 Americans have a transplanted kidney, but fewer than 17,000 per year will receive a transplant, despite a waiting list of 122,000. About 12 people a day in the U.S. die waiting for a kidney transplant.
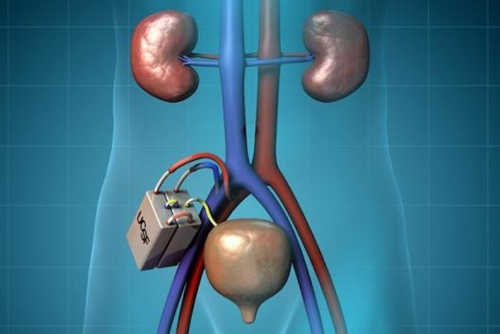
The artificial kidney implant is meant to give an alternative to dialysis for people with failed kidneys as they await a transplant. The new project brings together work at UC-San Francisco led by bioengineering professor Shuvo Roy, and nephrologist/engineer William Fissell at Vanderbilt, on the school’s medical and bioengineering faculties. Roy, Fissell, and other colleagues aim to apply advances in microelectromechanical systems and nanotechnology to construct a self-contained miniaturized device that resides inside the recipient’s body.
UC-San Francisco’s Kidney Project, directed by Roy, is working on key components of the proposed device. One of those elements is a silicon filter for removing waste products from blood, which with nanoscale pores in the silicon membrane can provide more uniform filtering than now offered in conventional dialysis machines.
Another key component is a bioreactor with tubule cells from human kidneys that perform metabolic functions needed to maintain healthy pH and potassium levels in the blood. A version of the bioreactor in an external device, developed with collaborator David Humes at University of Michigan, when combined with finer-grained filtration, increased survival rates of kidney failure patients in a hospital intensive care unit, compared to dialysis alone.
Roy, Fissell, and colleagues plan to use non-reactive coatings on the implanted device, which the researchers say will remove a need for patients to take drugs to suppress their immune systems. Tests so far show the bioreactor can last up to 60 days under simulated conditions. Early preclinical tests of the silicon filters will test their performance and ability to stay clot-free for 30 days. The grant is expected to be divided about equally between optimizing the filters and the bioreactor.
While the artificial kidney implant device is still in development, it is already designated for review by the Food and Drug Administration under the agency’s Expedited Access Pathway program. That program is designed to speed review of medical devices that address unmet needs for life threatening or debilitating diseases. FDA seeks to reduce the time and cost of review of review for designated devices, without compromising standards of safety or effectiveness.
Roy and Fissell have ownership stakes in Silicon Kidney LLC, a start-up enterprise in San Francisco that aims to commercialize the silicon membrane technology developed in the artificial kidney project.
Read more:
- Heart-Powered Leadless Pacemaker in Development
- Chip Device in Development to Simulate Human Gut
- Manufacturing Process Devised for Skin-Patch Electronics
- Hydrogel Aids Stem Cells Repair Heart Functions
- 3-D Printed Guide Devised to Regrow Nerve Fibers
* * *

 RSS - Posts
RSS - Posts
You must be logged in to post a comment.