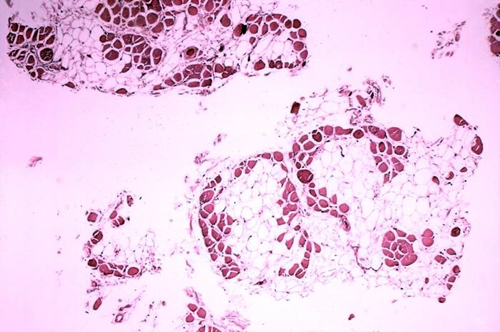
Cross-section of muscle tissue from a person with Duchenne muscular dystrophy shows extensive replacement of dark colored muscle fiber with light-colored adipose or fat cells. (Centers for Disease Control and Prevention)
8 January 2015. Grünenthal Group, a German pharmaceutical company, is licensing a treatment candidate for Duchenne muscular dystrophy developed by Akashi Therapeutics, a biopharmaceutical company created by organizations advocating for research on the disease. While full financial details of the agreement were not disclosed, Grünenthal says it’s committing more than $100 million to the venture.
Duchenne muscular dystrophy is a rare genetic disorder resulting in progressive muscle degeneration and weakness, primarily in the shoulders, arms, hips, and thighs. The disease affects mainly boys starting at age 3 to 5, and caused by a defective gene that fails to produce the protein dystrophin for strengthening muscle fiber and protecting muscles from injury. While life expectancy can vary, people with Duchenne muscular dystrophy do not often survive past their 20s or 30s, with death caused by respiratory or cardiac failure.
The deal involves Akashi’s drug candidate code-named HT-100. Akashi Therapeutics, in Cambridge, Massachusetts was founded by patient advocacy organizations Charley’s Fund and Nash Avery Foundation expressly to find therapies for Duchenne muscular dystrophy. HT-100 is a delayed release formulation of halofuginone, a derivative the compound febrifugine found in the roots of a Chinese hydrangea plant known as chang sham, long associated with herbal treatments for malaria, fibrosis, and inflammatory diseases.
Akashi designed HT-100 as a small-molecule candidate to reduce fibrosis and inflammation, as well as regenerate healthy muscle fiber in patients with Duchenne muscular dystrophy. HT-100 is in an intermediate-stage clinical trial with 30 participants at 4 sites in the U.S. The drug is designated an orphan therapy by regulators in both the U.S. and Europe, and is granted fast-track review status in the U.S.
Up to recently, Grünenthal Group in Aachen, Germany developed treatments solely for pain and inflammatory disorders, but broadened its focus in 2014 to niche markets addressing unmet medical needs. Under the agreement, Grünenthal is providing Akashi with unspecified initial and milestone payments, and will fund development and commercialization beyond intermediate-stage clinical trials. Akashi will also be eligible for royalties on net sales in the U.S. Grünenthal says it plans to put more than $100 million into the initiative.
Akashi is also working on a muscle-building compound code-named DT-200, which the company says is ready for early-stage efficacy trials. In addition, Akashi is developing AT-300, a modified peptide derived from the venom of the Chilean Rose Tarantula that targets calcium imbalance in muscles, considered an early trigger of later weakening and loss of muscle function. In 2010, Science & Enterprise reported on development of this therapy at University at Buffalo, which was licensed to Tonus Therapeutics, a start-up company for development. Akashi acquired the technology from Tonus in 2014.
Read more:
- Advocacy Group, Genetics Company Partner on Brain Disorder
- Gene Editing Licensed for Inherited Disease Treatments
- Evotec, Foundation Partner on Rare Juvenile Disease
- Hemophilia Therapy Candidate Given Breakthrough Status
- Patient Registry Started for Rare Genetic Disorder
* * *

 RSS - Posts
RSS - Posts
You must be logged in to post a comment.