1 September 2016. Takeda Pharmaceutical Company is receiving $19.8 million from a U.S. public health agency for initial steps to develop a vaccine to prevent Zika infections. Work to develop the vaccine could earn Takeda, in Osaka, Japan, as much as $312 million if the Biomedical Advanced Research and Development Authority or BARDA, in the U.S. Department of Health and Human Services, exercises all options with the company including late-stage clinical trials and filing a biologics drug application at the Food and Drug Administration.
The Zika virus is transmitted primarily by aedes aegypti mosquitoes, the same species carrying chikungunya, dengue, and yellow fever pathogens. The virus may also be spread through sexual contacts. Most people contracting the Zika virus report symptoms such as mild fever, conjunctivitis or pink eye, and muscle and joint pain. The current Zika outbreak, however, is resulting in increasing numbers of cases of birth defects, notably microcephaly and Guillain-Barré syndrome.
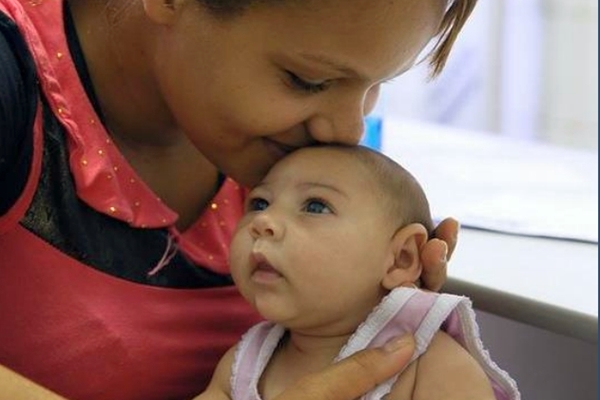
Microcephaly is a rare neurological condition where the baby’s head is noticeably smaller in size, with the children often experiencing developmental issues. Guillain-Barré syndrome is an autoimmune disorder, where the immune system attacks part of the peripheral nervous system, leading to progressive muscle weakness and eventual paralysis. The paralysis can become life-threatening if it affects breathing and heart muscles.
Centers for Disease Control and Prevention reports as of 31 August 2016, some 2,700 Zika cases in U.S. states and more than 14,000 cases in U.S. territories, with 624 pregnant women showing evidence of Zika infection in U.S. states and another 971 pregnant women with Zika infections in U.S. territories. There are currently no treatments for Zika infections, nor is there a vaccine to prevent infections.
The agreement with BARDA calls for Takeda to develop a vaccine for Zika with an inactivated virus, as well as adjuvants or boosters to increase its potency. The current funding covers initial research and development and preclinical studies leading to a vaccine candidate submitted in an investigational new drug application to FDA. The funding also covers an early-stage clinical trial of the vaccine by 2017. Takeda’s facilities in Hikari, Japan will manufacture the vaccine.
Rajeev Venkayya, president of Takeda’s vaccines division, says in a company statement that “The Zika emergency demands swift action by governments, public health agencies, medical and scientific communities, industry, and others, and partnerships are essential for success.” The company says its work with vaccines goes back 70 years and includes vaccines for dengue, norovirus, and polio.
Read more:
- PhRMA: 258 Vaccines in Clinical Development
- Molecular Express, Baylor Medicine Partner on Vaccines
- Common Antibodies Found for Multi-Virus Flu Vaccine
- RNA Technology Allows for Fast Vaccine Development
- Bacterial-Biopolymer Delivery System Designed for Vaccines
* * *

 RSS - Posts
RSS - Posts
You must be logged in to post a comment.