25 May 2018. The U.S. Food and Drug Administration authorized marketing of software to help diagnose wrist fractures using machine-learning algorithms to read and interpret X-ray images. FDA’s action clears the software known as OsteoDetect, made by Imagen Technologies in New York City, for sale and distribution in the U.S.
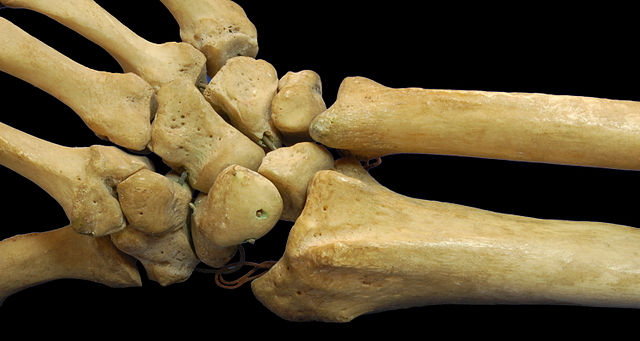
Imagen makes software for medical practitioners, which according to the company’s LinkedIn page, “is creating a world without diagnostic errors.” In this case, the software helps diagnose a distal radius fracture in the wrist, considered the most commonly broken bone in the arm. A distal radius fracture can occur when an outstretched arm tries to break a fall. Older people and those with osteoporosis are susceptible to these fractures, often when falling from a standing position. But a distal radius fracture can also occur in younger people with healthy bones with enough force, usually from an accident or other trauma.
OsteoDetect, according to FDA, reads X-ray images of distal radius fractures in adults from two dimensions, front-to-back and from the sides. Its machine-learning algorithms identify suspected sources of breaks in the bones, and highlight the suspected breaks for physicians. The software’s conclusions are meant to be advisory, says FDA, and are not designed to replace the judgement of a physician. Nonetheless, the software is intended for use in primary care, emergency medicine, urgent care, and specialty practices, such as orthopedics.
FDA says it granted the marketing clearance for OsteoDetect based on results of studies supplied by Imagen Technologies. One of the studies used OsteoDetect to evaluate 1,000 X-ray images of wrist fractures, and identify the site of the fractures, compared to the judgements of 3 board-certified orthopedic surgeons. In a second study, 24 providers reviewed the software’s recommendations made on 200 patient cases. In both studies, says FDA, OsteoDirect helped physicians detect wrist fractures faster and more accurately, compared to the physicians working without the software.
Authorization of OsteoDetect was made under FDA’s de novo premarket review pathway, assigned for devices considered to have low to moderate health risks, and in cases where there are no equivalent devices already cleared for marketing. Imagen Technologies is a 3 year-old company that operates largely under the radar. Its web site makes no mention of OsteoDirect or any other specific product. The industry web site Mass Device says Imagen raised $14.3 million in equity financing in March 2017.
More from Science & Enterprise:
- Virtual Biopsy in Development to Detect Melanoma
- Wearable Devices Explored to Detect Emotional States
- FDA Clears A.I. Software Detecting Diabetic Eye Disease
- Self-Service A.I. Offered for Pathologists
- A.I./Image Analysis Diagnose Eye, Lung Diseases
* * *

 RSS - Posts
RSS - Posts
You must be logged in to post a comment.