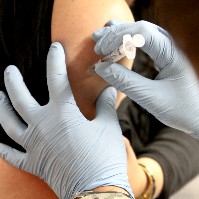
The U.S. Department of Health and Human Services’ Biomedical Advanced Research and Development Authority (BARDA) announced the first contract awards to upgrade the infrastructure for producing medical countermeasures that protect against natural and man-made biological threats.
The eight contracts are expected to reduce the time and cost of development, testing, and production of medical countermeasures and improve the safety, efficacy, and ease of use of these products. The awards total $55 million for the initial phase and up to $100 million over three years.
The contracts cover the following work …
- VaxDesign will further develop its MIMIC platform, an in vitro (test-tube-based) human immune system mimetic designed to accelerate evaluation of candidate and stockpiled vaccine safety and effectiveness by supplementing animal testing.
- PATH will test multiple formulation chemistries and strategies to increase the shelf-life of influenza vaccines, which has implications for the national vaccine stockpile as well as cold-chain requirements domestically and in developing countries.
- IDRI will develop and evaluate adjuvant formulations to enhance influenza vaccine immunogenicity and cross-protection to make them more effective against novel viral strains that may cause the next pandemic.
- Pfenex will apply its Pfenex Expression Technology Platform to the development of bioprocesses for production of a stable candidate anthrax vaccine.
- Novartis Vaccines and Diagnostics will by investigate techniques for the development of optimized influenza seed virus, a critical issue to begin manufacturing of influenza vaccine against a newly identified strain
- Rapid Micro Biosystems will address a critical issue for release of influenza vaccine after manufacturing: development of methods for accelerated sterility testing. These improvements in production processes could shave weeks off the influenza vaccine manufacturing and product release schedule.
- 3M and Northrop Grumman will develop rapid, high-throughput surveillance and molecular diagnostics.
Related: HHS Review Highlights Role of Business in Medical Countermeasures

 RSS - Posts
RSS - Posts
[…] This post was mentioned on Twitter by Alltop Science, Rada and Juliet, Alan Kotok. Alan Kotok said: Contracts Awarded for Countermeasure Upgrades | #Science #Business http://t.co/pTtEnH8 #ScienceBusiness […]
[…] Read more: Contracts Awarded for Countermeasure Upgrades […]
[…] Read more: Contracts Awarded for Countermeasure Upgrades […]
[…] Read more: Contracts Awarded for Countermeasure Upgrades […]