25 Sept. 2020. Dexcom Inc., a maker of glucose monitoring systems, and University of Virginia are collaborating on extending the company’s current technologies to cover more types of diabetes. Financial aspects of the five-year agreement were not disclosed.
Diabetes is a chronic disorder where the pancreas does not create enough insulin to process the sugar glucose to flow into the blood stream and cells for energy in the body. In type 2 diabetes, which accounts for at least 90 percent of all diabetes cases, the pancreas produces some but not enough insulin, or the body cannot process insulin. According to the International Diabetes Federation, diabetes affects an estimated 463 million people worldwide, of which 51 million are in North America.
People with type 1 diabetes have an inherited autoimmune disorder where beta or islet cells in the pancreas do not produce insulin. Type 1 diabetes is diagnosed primarily in children or young adults, where the immune system is tricked into attacking healthy cells and tissue as if they were foreign invaders, in this case, insulin-producing islet cells.
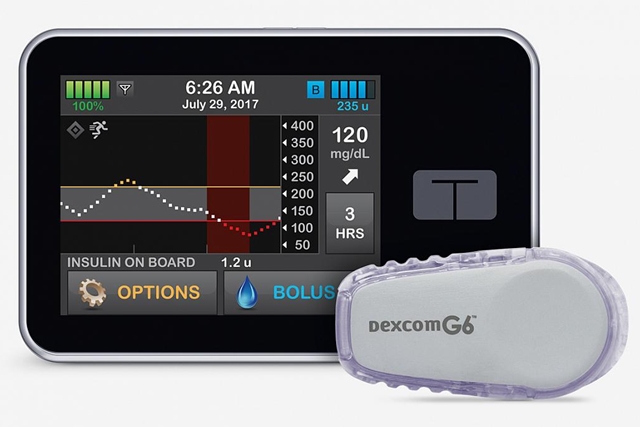
Dexcom, in San Diego, develops systems that monitor a person’s blood glucose levels, and when combined with a self-contained or closed-loop dispensing system, can automatically release insulin into an individual’s blood stream. In January 2020, as reported in Science & Enterprise, FDA cleared a so-called artificial pancreas system for people with type 1 diabetes that combines Dexcom’s continuous glucose monitoring, or CGM, systems with an insulin pump made by Tandem Diabetes Care, also in San Diego.
The Dexcom/Tandem system is controlled with algorithms designed at the Center for Diabetes Technology at University of Virginia in Charlottesville. The algorithms were licensed by the university to a spin-off enterprise TypeZero Technologies LLC, also in Charlottesville, founded by Center for Diabetes Technology researchers Boris Kovatchev and Marc Breton. In 2018, DexCom acquired TypeZero Technologies.
Under the new agreement, DexCom and UVa will partner on extending the company’s current glucose monitoring and insulin delivery technologies to cover individuals with the more prevalent type 2 diabetes, hospitalized patients, and other forms of the disease, such as gestational diabetes affecting pregnant women. The work includes refinements of algorithms for controlling the devices, originally written at UVa, for eventual testing in clinical trials. DexCom says it plans to expand its Charlottesville operations, established with the TypeZero acquisition.
Breton and Kovatchev at the Center for Diabetes Technology are expected to lead the UVa research team, which will bring in faculty colleagues from the university’s medical, data science, and engineering schools.
Kevin Sayer, president and CEO of Dexcom, says in a company statement released through BusinessWire, “We are investing heavily in research and development efforts that explore new diagnostic tools and treatments. By partnering with a top-tier research institution like the University of Virginia, we hope to bring better CGM solutions to patients much faster than we could alone.”
More from Science & Enterprise:
- Phone Camera, A.I. Detect Early Diabetes
- Vaccine Blocks Suspected Type 1 Diabetes Infections
- Engineered Antibody Delays Type 1 Diabetes Onset
- Infographic – Highest Diabetes Rates in U.S.
- Small Business Grant Funds Diabetes Rescue Device
* * *

 RSS - Posts
RSS - Posts
You must be logged in to post a comment.