23 Nov. 2020. A new company is underway creating off-the-shelf engineered cells from the immune system as cancer treatments, and raising $42 million in venture funds. Catamaran Bio Inc., in Cambridge, Massachusetts, is spun-off from medical school research labs at University of Minnesota and George Washington University.
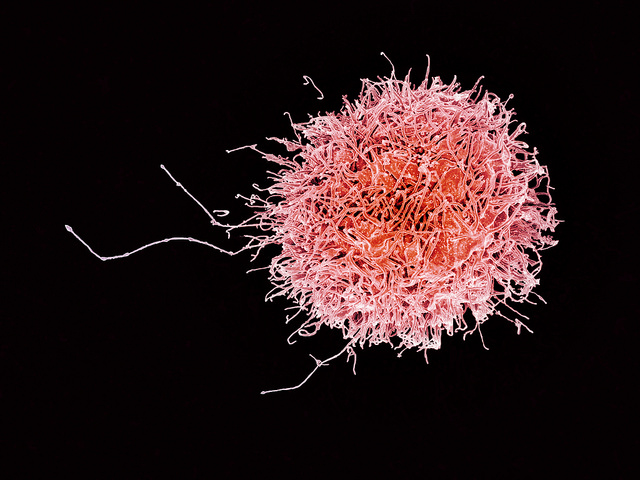
Catamaran Bio is developing treatments for cancer based on natural killer cells in the immune system. Like their cousins B- and T-cells, natural killer cells are white blood cells, which act against cells infected by viruses and in early-stage tumors. Catamaran’s technology, called Tailwind, alters donated natural killer cells to express chimeric antigen receptors, or CARs, proteins attracting antibodies that bind to and destroy blood-related and solid tumor cancer cells.
The company says its CAR natural killer or CAR-NK cells are designed for signaling inside cells, which maximizes production of immune-system enzymes aimed at tumor cells. Catamaran says its CAR-NK cells are also programmed to sense and evade the microenvironment that surrounds and supports tumors, as well as suppresses immune system activity against tumors. In addition, says the company, CAR-NK cells can carry larger genetic payloads, increasing their ability to deliver complex, multi-stage cancer treatments. And the company’s therapies are derived from natural killer cells from healthy donors, engineered to be administered as allogenic, or off-the-shelf treatments without relying on the patients’ own cells.
The company says it has two lead cancer treatments, one each for solid tumors and blood-related cancers, still in the discovery/lead-optimization stage. Both CAR-NK therapies, says Catamaran, targets the same antigen that appears on the surface of cancer cells. The company says its preclinical studies indicate the lead treatments show effects against both solid tumor and blood-related cancers, but the company so far is concentrating on solid tumors.
Catamaran Bio’s CAR-NK technology is based on research from the labs of its scientific founders, Branden Moriarity, professor of pediatrics and hematology/oncology at University of Minnesota, and Catherine Bollard, professor of pediatrics, microbiology, immunology, and tropical medicine at George Washington University. Bollard is also on the faculty at Children’s Research Institute, Children’s National Hospital in Washington, D.C. Moriarty and Catamaran are collaborating on natural killer cell engineering in Moriarty’s lab at Minnesota.
“Catamaran is focused on expanding the frontier of cell therapies to treat solid tumors and provide transformative benefit to cancer patients,” says Vipin Suri, Catamaran Bio’s chief scientist in a company statement. “We are doing this by creating allogeneic cell therapies that harness the innate cancer-fighting power of NK cells and enhancing them with new biologically-powerful attributes from our leading-edge technologies, all originating from our custom-built Tailwind platform for designing, engineering, and manufacturing off-the-shelf CAR-NK cell therapies.”
The company was founded earlier this year, and is now raising $42 million in its first venture funding round. Sofinnova Partners in Paris and Lightstone Ventures in Menlo Park, California, both life science investors, led the financing. Taking part in the round are founding investor SV Health Investors, and the venture arms of drug makers Takeda and Astellas.
More from Science & Enterprise:
- Cancer Immune System Therapy Start-Up Raises $53M
- Start-Up Developing Engineered B-Cell Therapies
- Cancer Center, Biotech Partner on Off-the-Shelf T-Cells
- Precision Cancer Therapy Start-Up Gains $110M in Early Funds
- Gilead Acquires Cancer Therapy Biotech for $21B
* * *

 RSS - Posts
RSS - Posts
You must be logged in to post a comment.